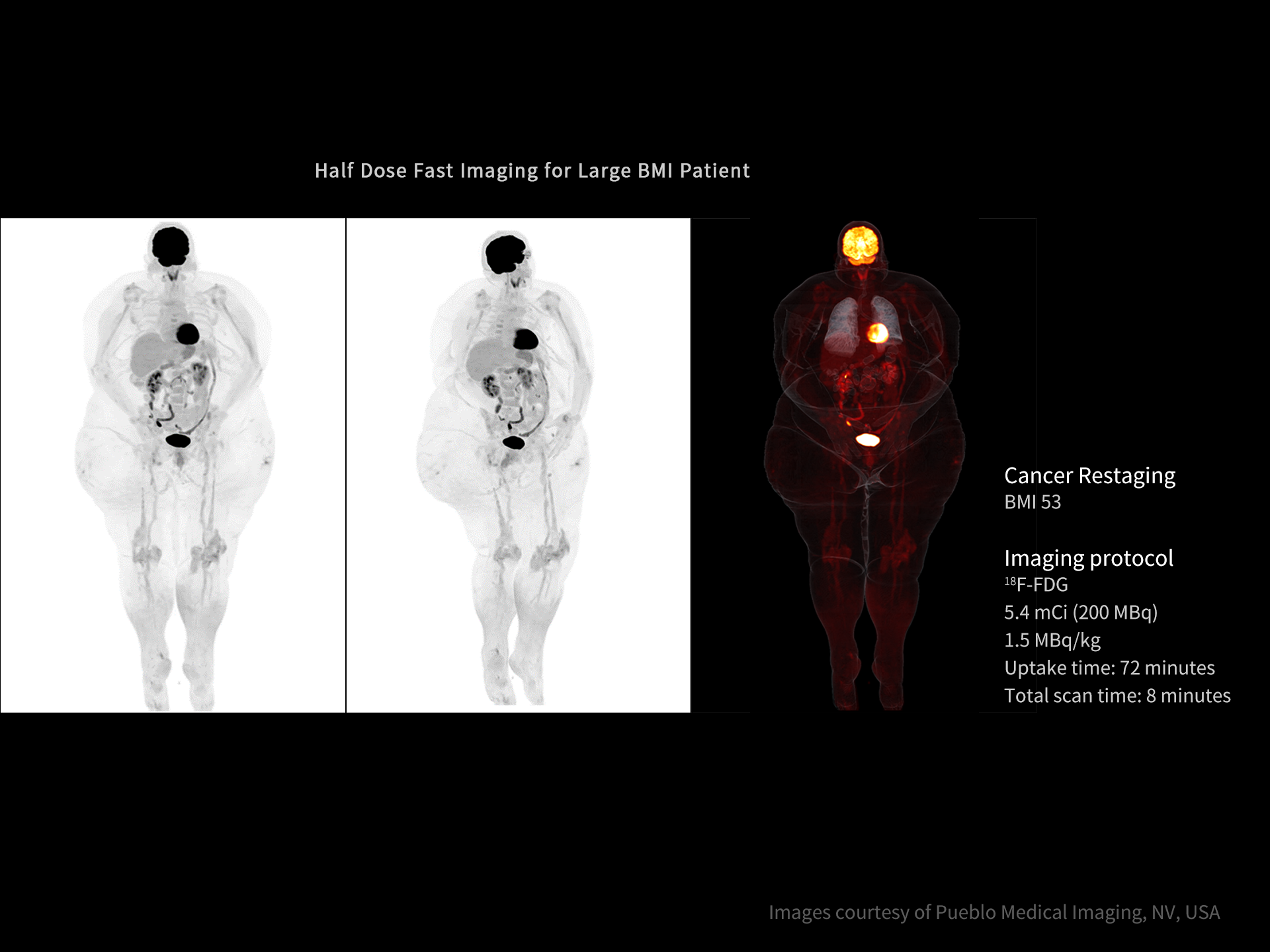
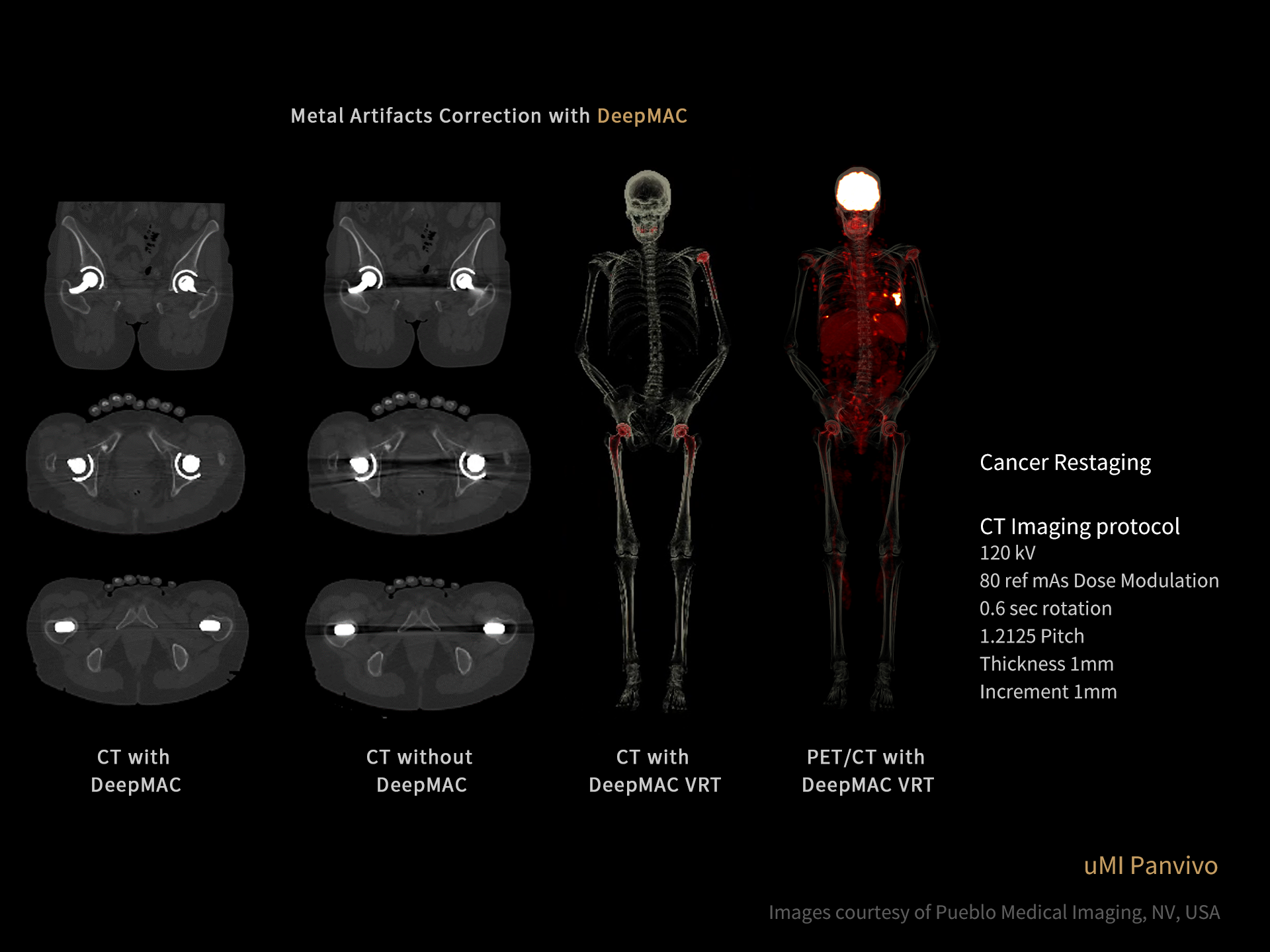
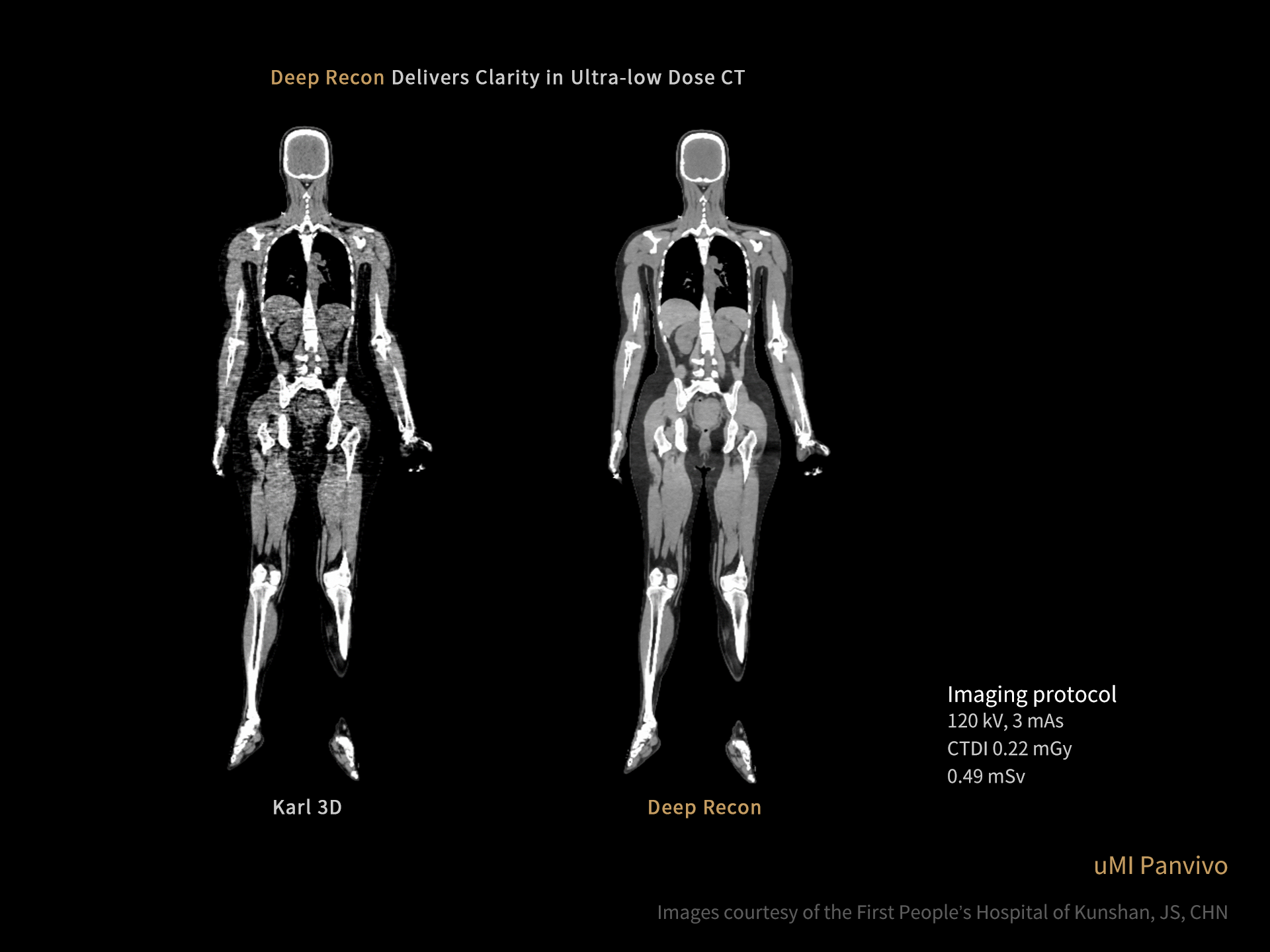
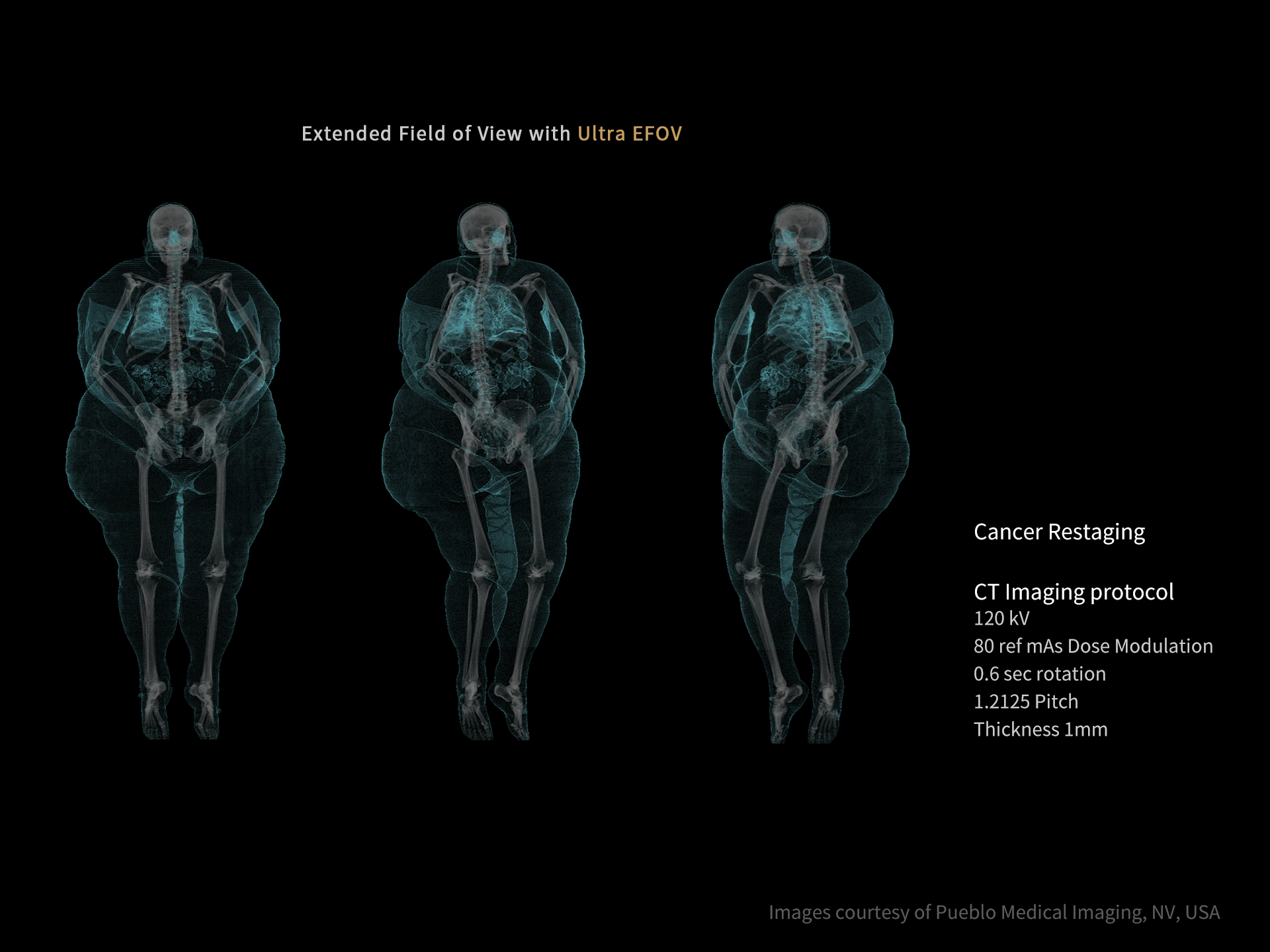
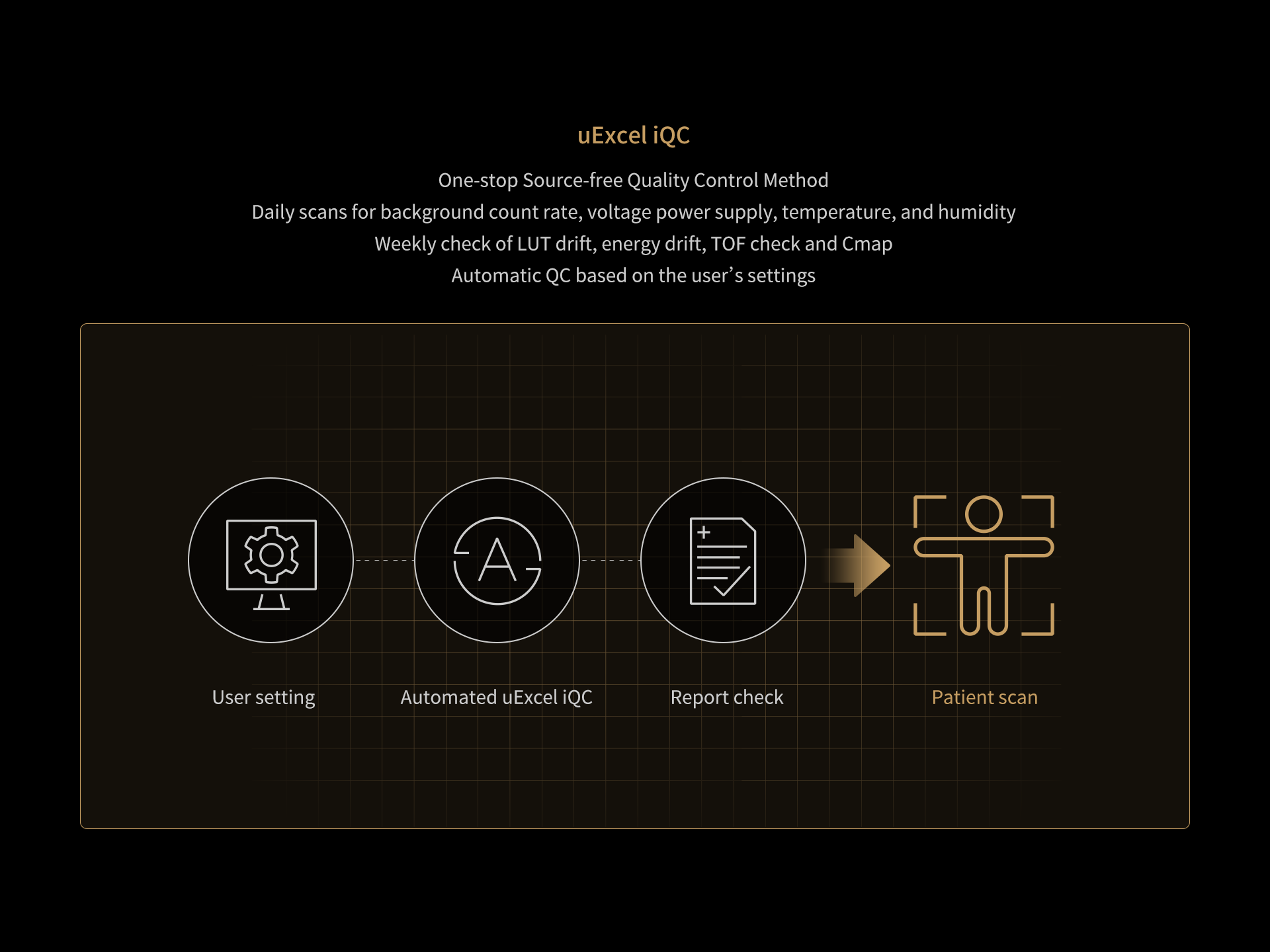
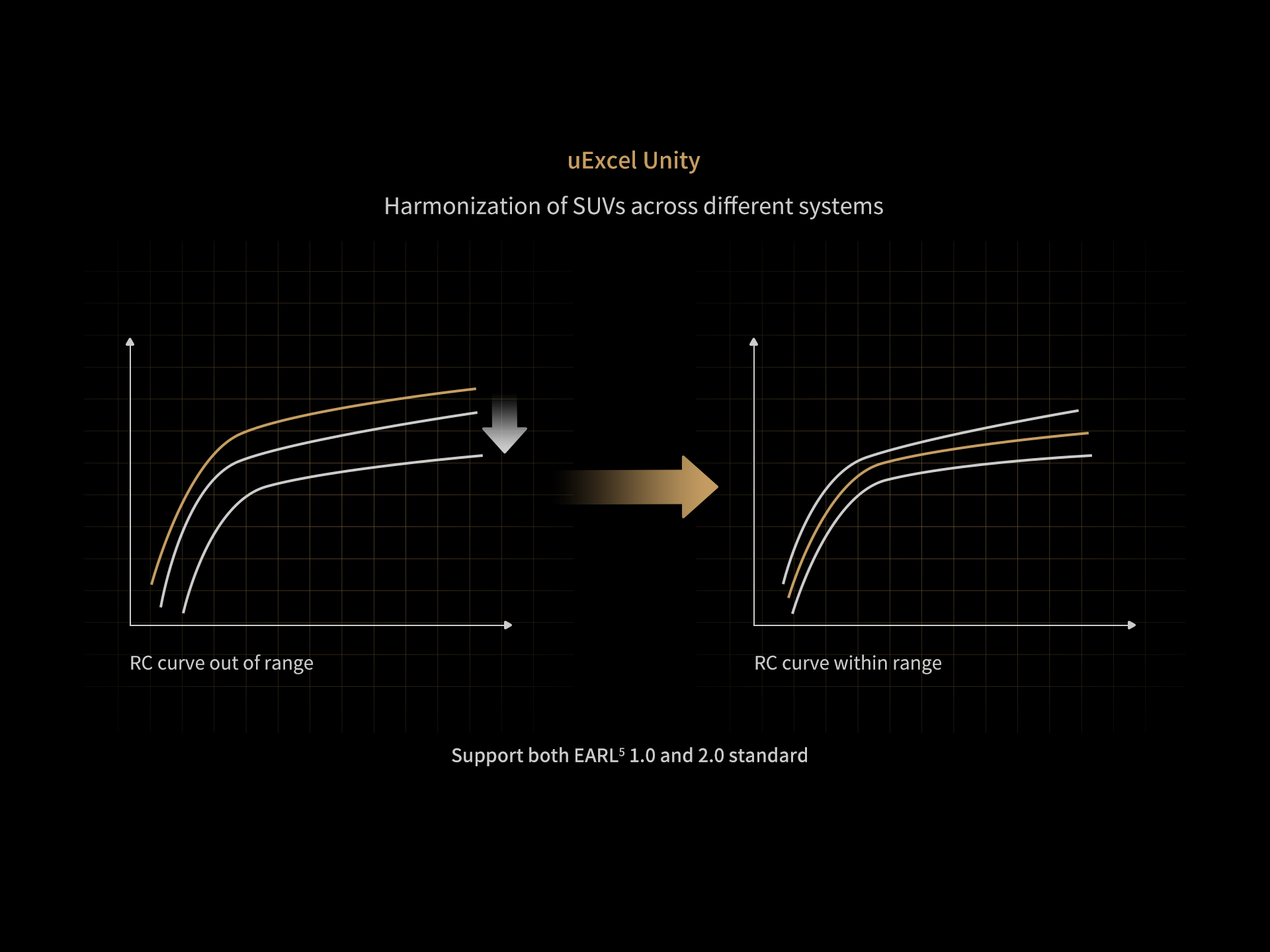
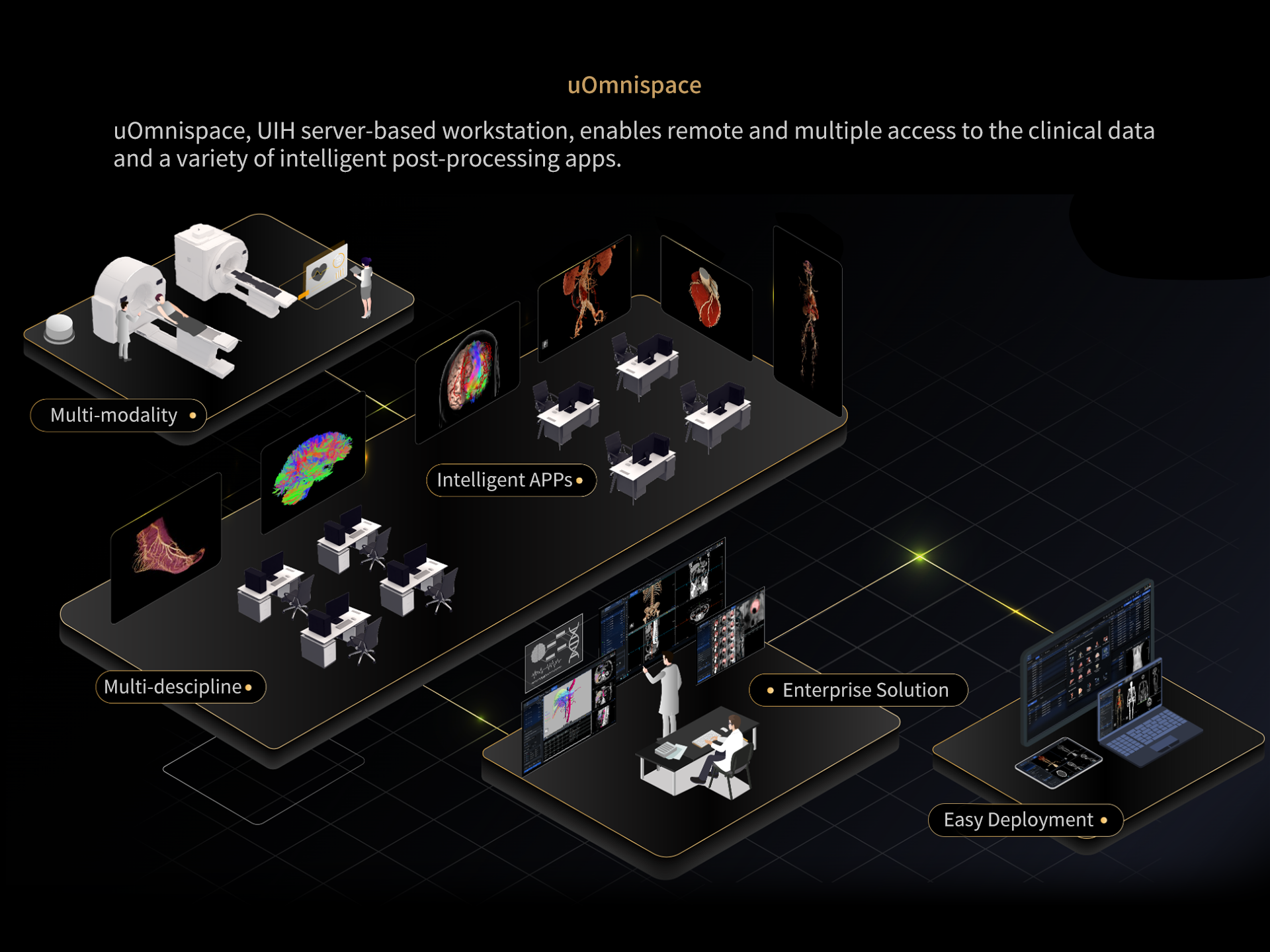
DISCLAIMERS
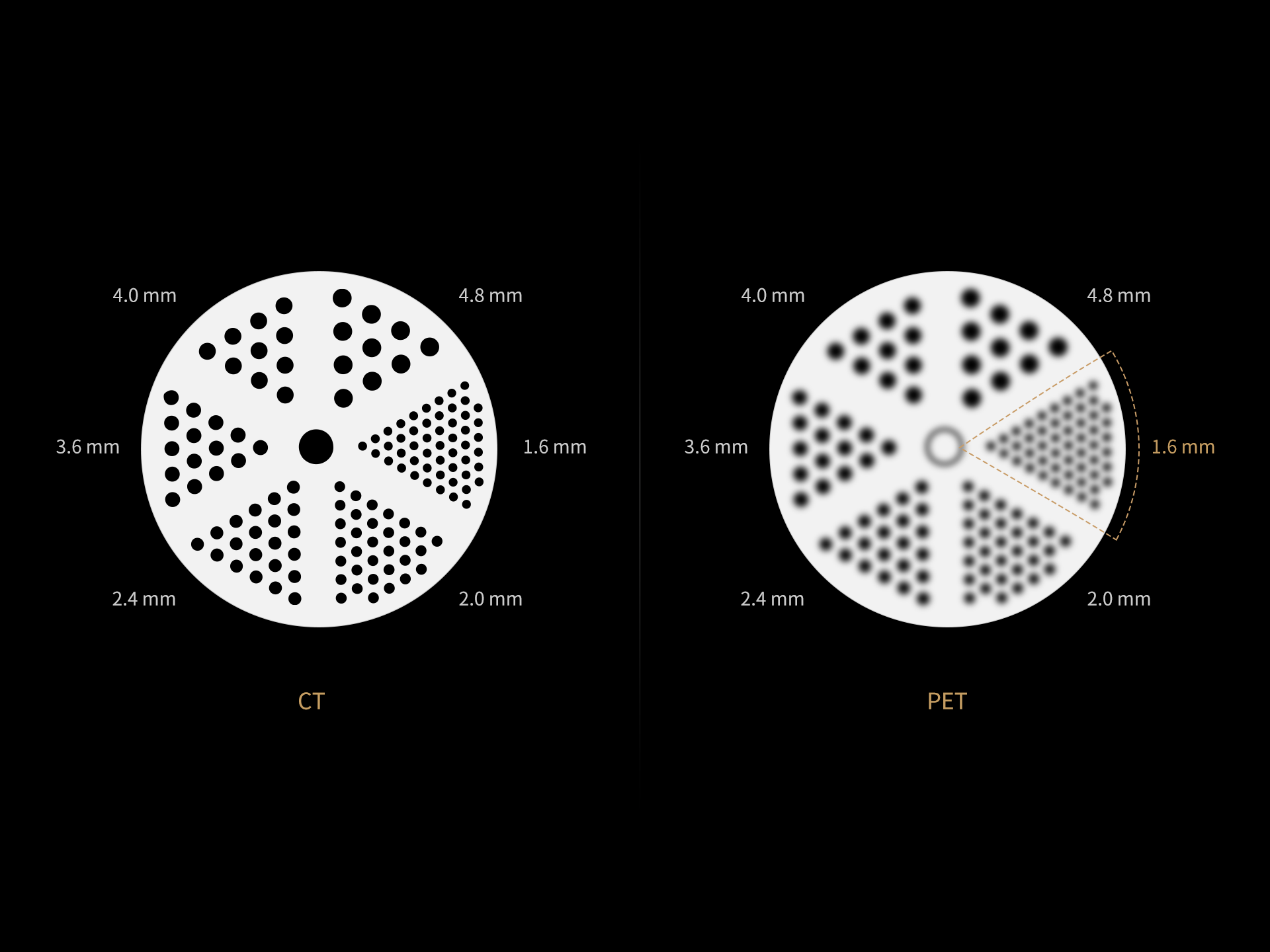
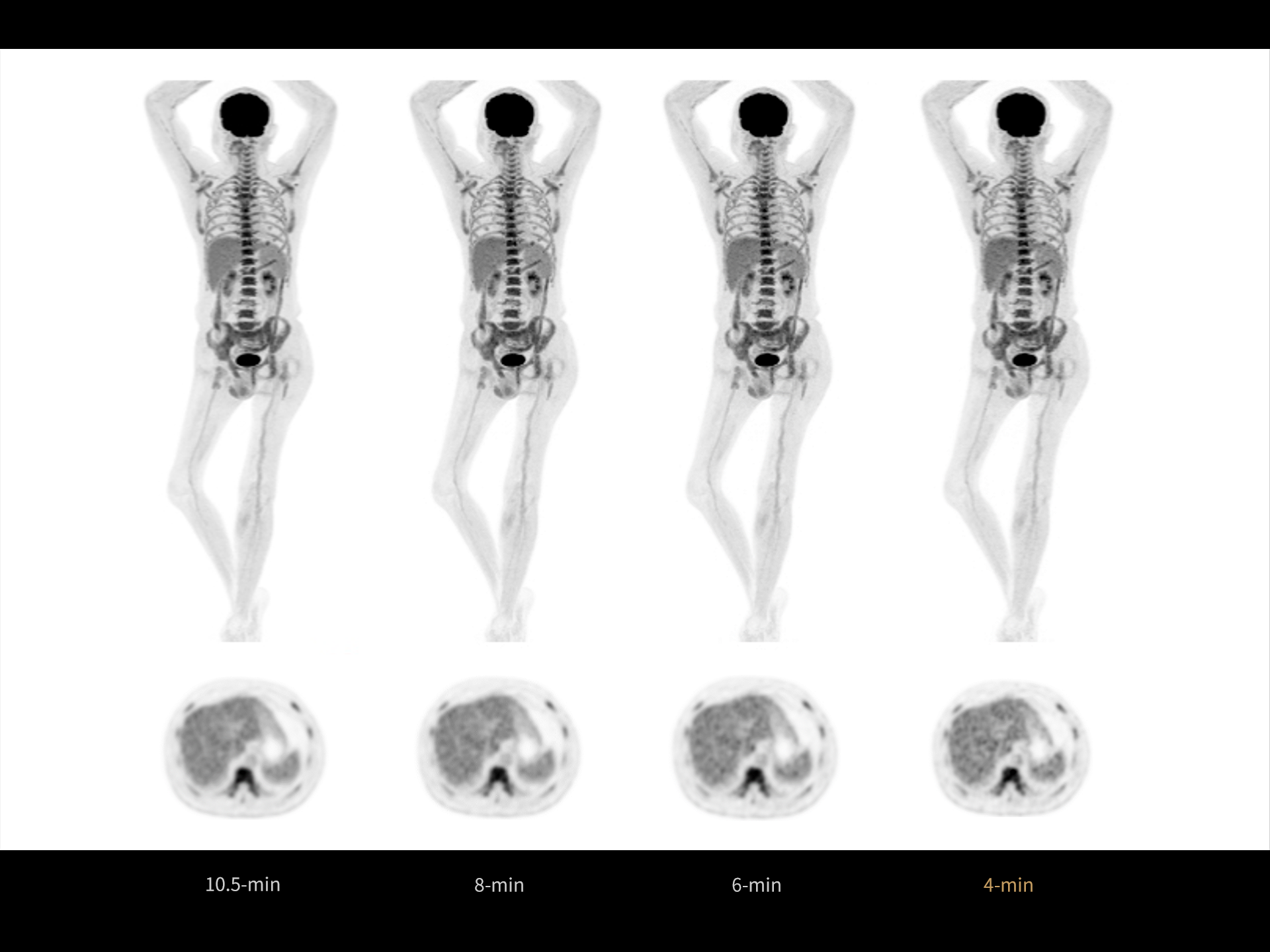
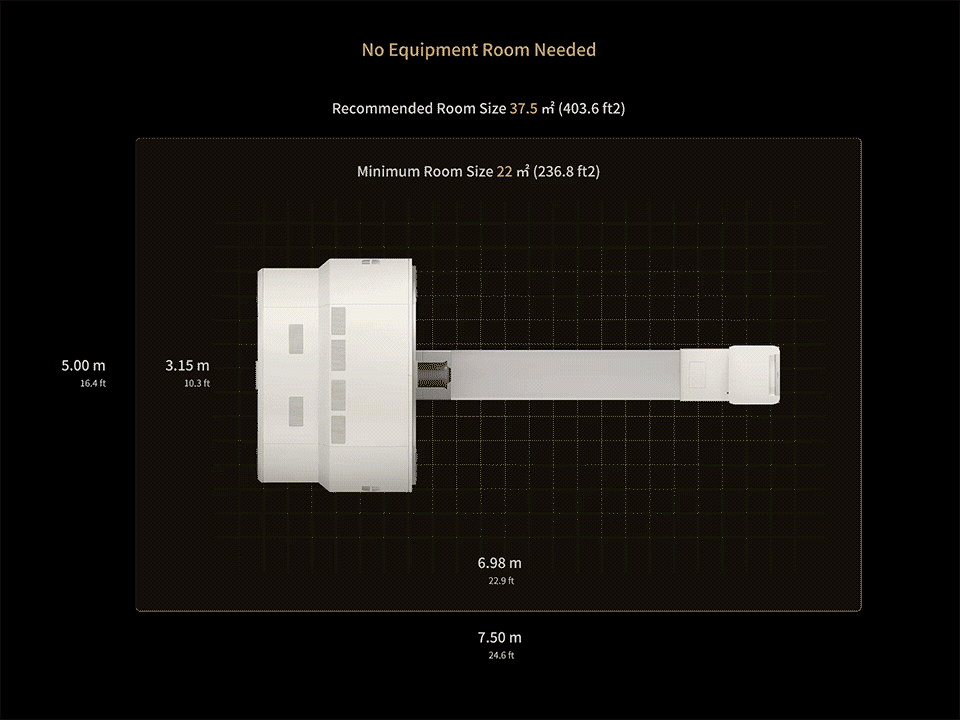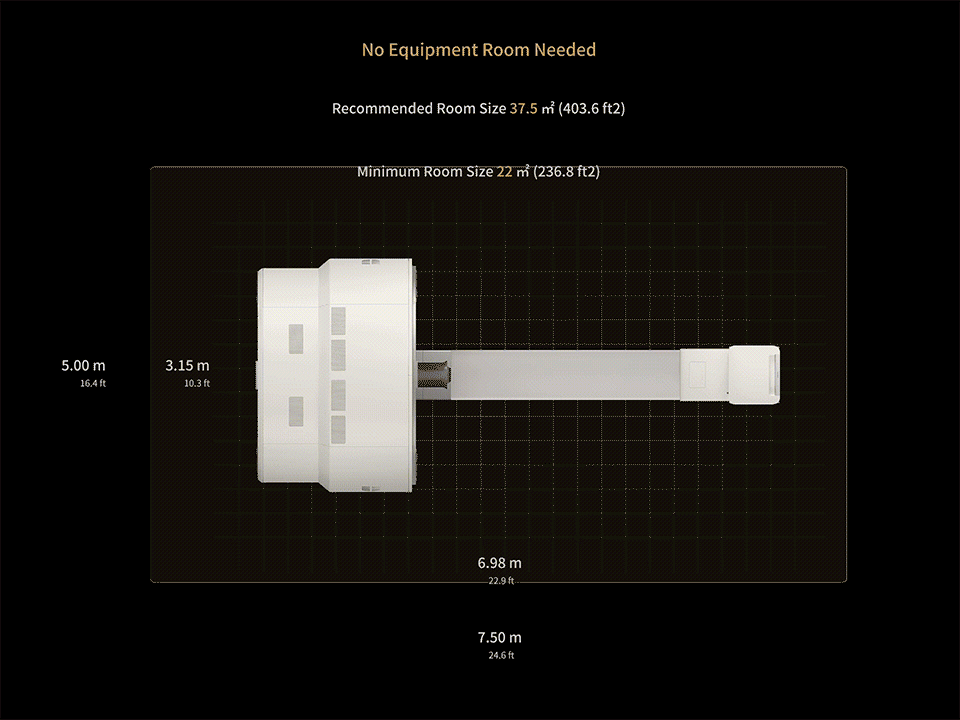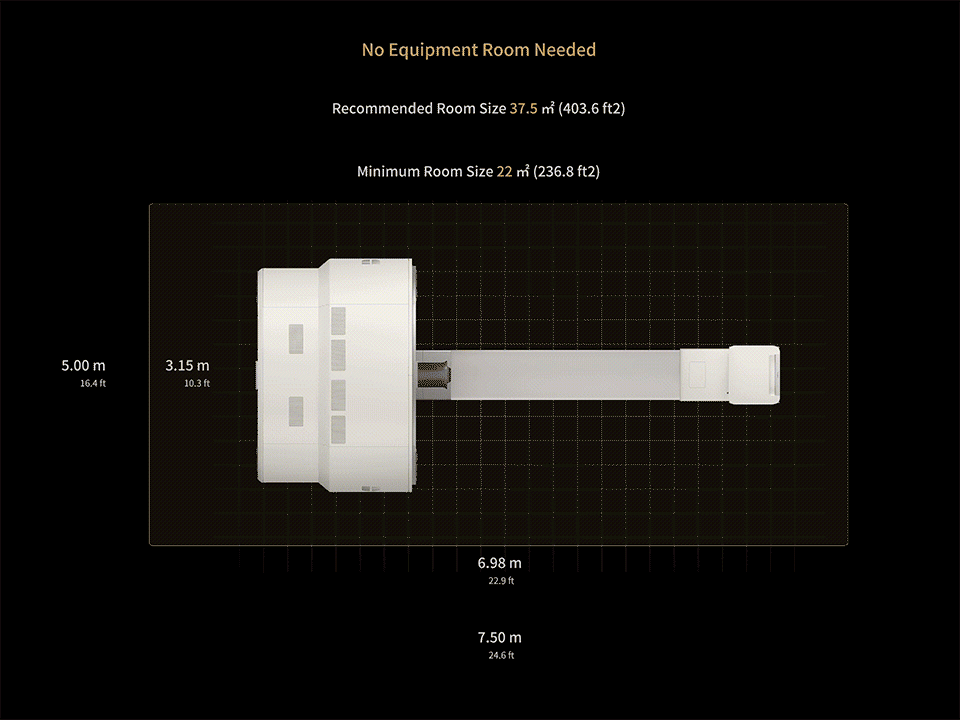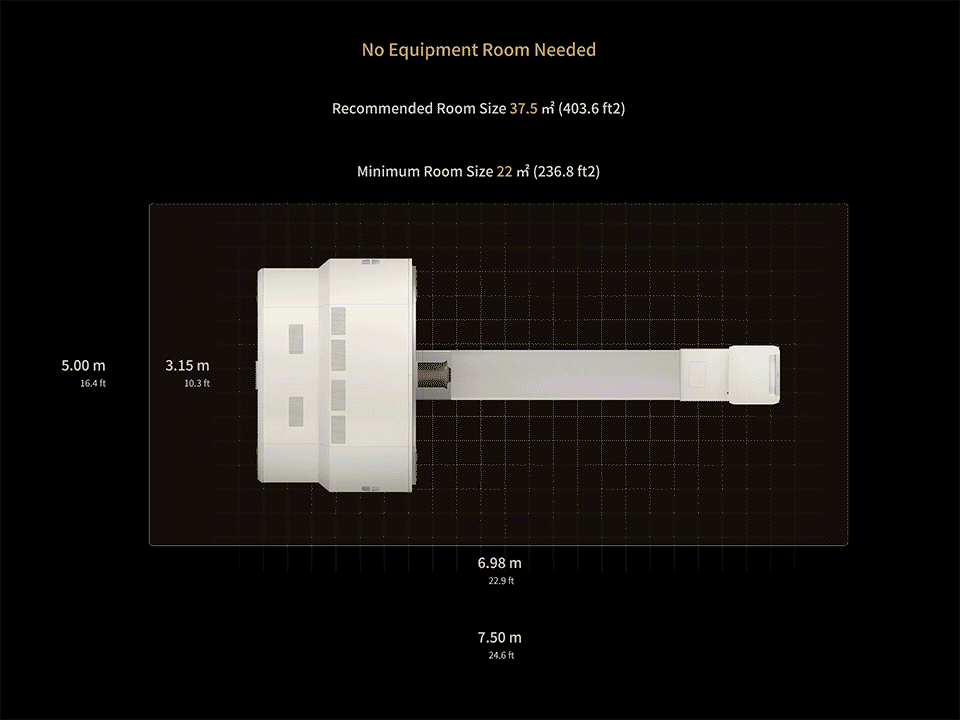
1. Data calculated with 35-cm object size.
2. uMI Panvivo LS (107 cm) is CE MDR cleared and FDA 510(k) pending.
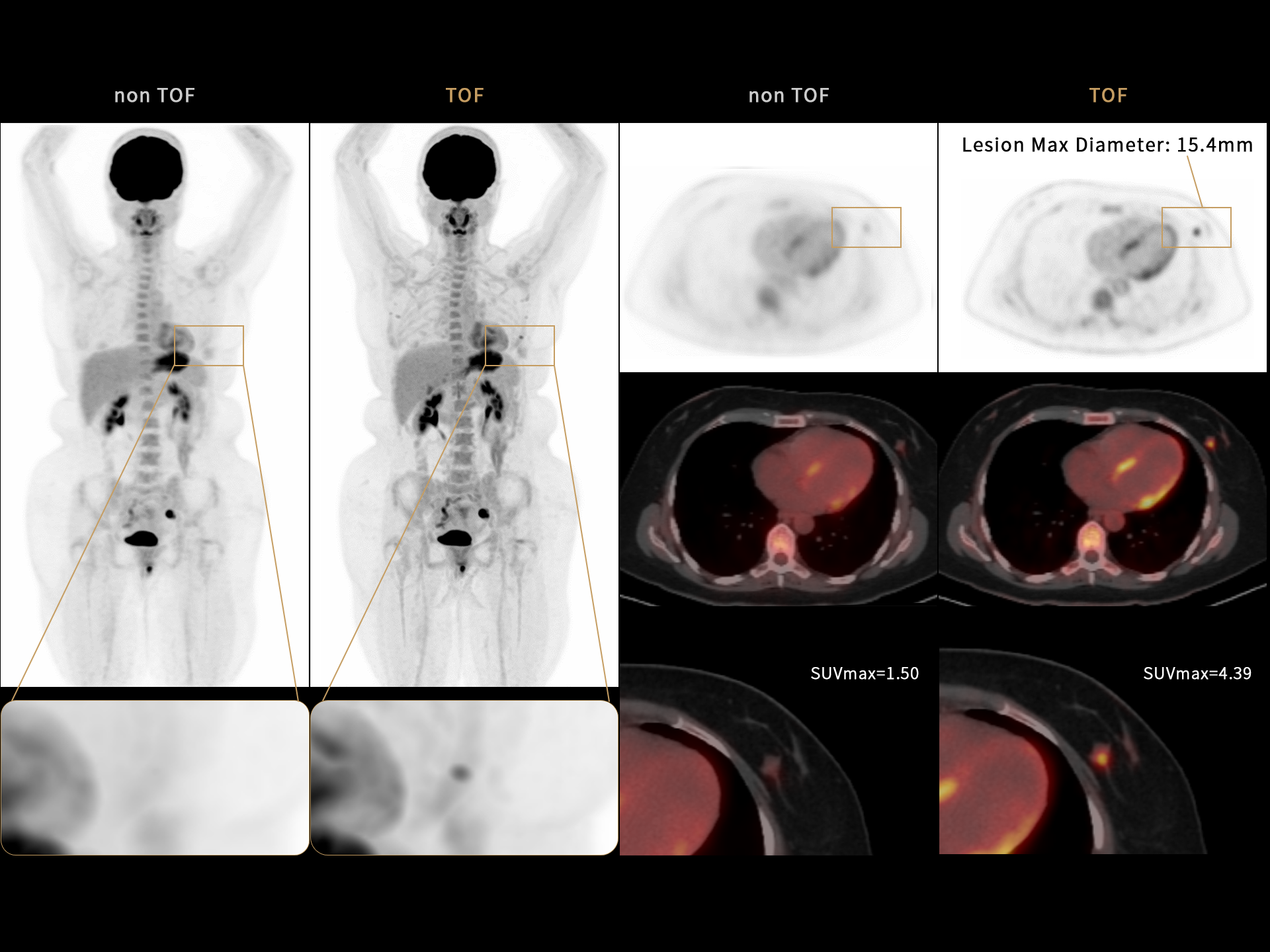
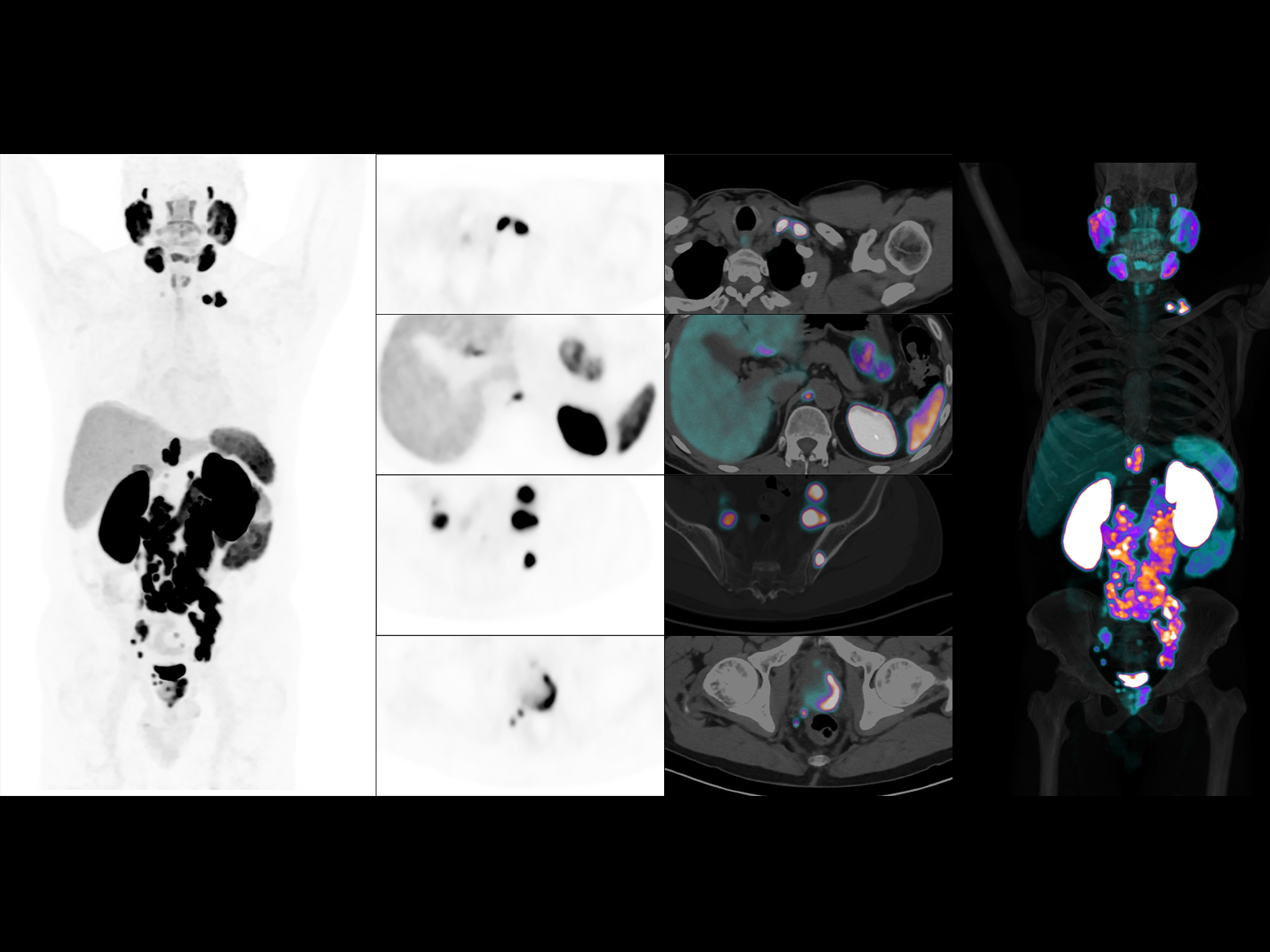
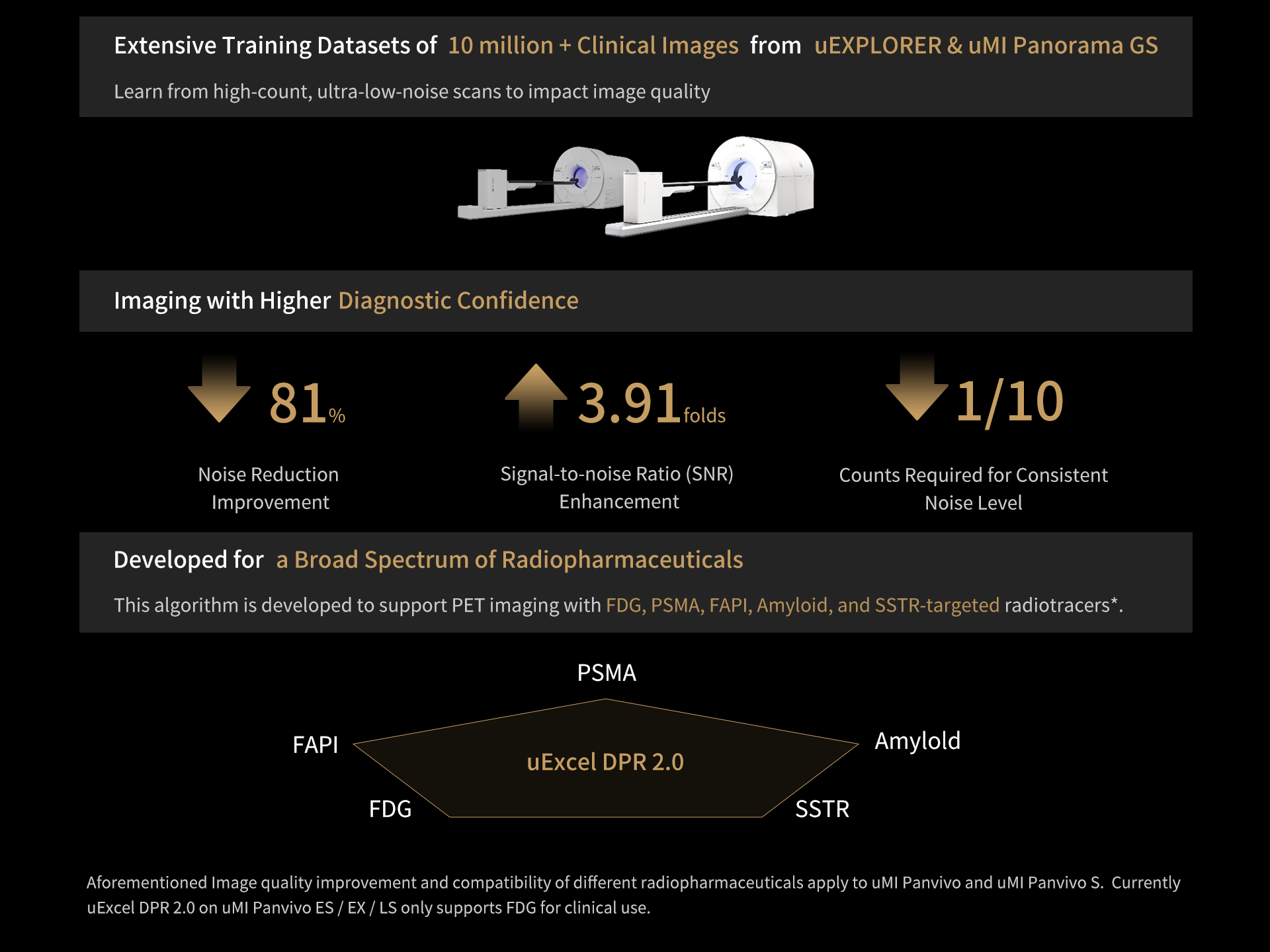
3. Aforementioned Image quality improvement and compatibility of different radiopharmaceuticals apply to uMI Panvivo and uMI Panvivo S. Currently uMI Panvivo ES and Panvivo EX only support FDG for clinical use. Currently uExcel DPR 2.0 on uMI Panvivo ES / EX / LS only supports FDG for clinical use.
4. uOmnispace can be deployed in conjunction with UII's PET-CT Oncology, which is not part of this product. Please visit uii-ai.com for more information on compliance, safety, and effectiveness of this application.
5. Earl: EANM Research Ltd.
6. uOmnispace is an independently registered product that is FDA 510(k) and CE MDR cleared.
The products/features are not commercially available in all countries. Their future availability cannot be guaranteed.
United Imaging does not make any claims regarding the safety and effectiveness of tracers that have not been approved by the FDA or CE.
Onscreen reproduction and display may result in a certain degree of image-quality degradation.